|
Ethyl mercaptan has two types of structures, called
rotamers, as shown in the figure to the
right. As the -SH thiol group part of the molecule rotates
around through 360 degrees, it passes through three minima. The most stable of these are two equivalent, mirror
image gauche conformers. The final, less stable minimum is a trans conformer, that occurs when the H on SH
faces away from the C–C–S backbone of the molecule. Spectral signatures of both the gauche and
trans conformers were found by the study described above, but there is better evidence that the gauche
conformer is present in Orion KL than the trans one. 77 lines in the survey match the spectrum of gauche
ethyl mercaptan.
Ethyl mercaptan's chemical cousin,
methyl mercaptan, was first reported in space
in 1979. Both compounds are added to combustible hydrocarbons as leak detectors due to their strong and
distinctive odors; ethyl mercaptan is the primary odorant
added to propane and butane.
|
|
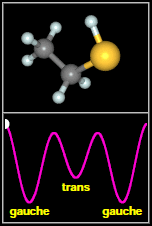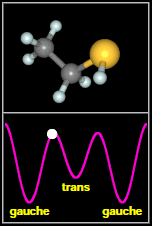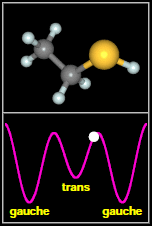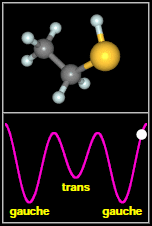
After Fig. 1(a,b) of Kolesniková et al.
|



