| |||
|
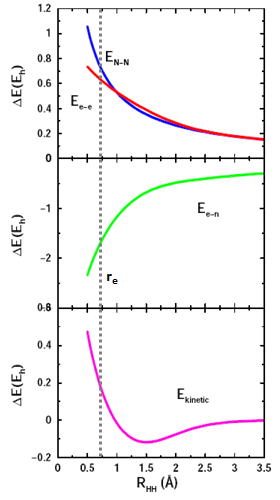
The following figure shows the contributions to the energy of H2 due to the new interactions between the two H atoms that increase as the atoms approach the minimum separation, re. | ||
|---|---|---|
| ||
|
As expected, the two electrons and the two protons in the nuclei each repel each other: Ee-e and EN-N both increase as the nuclei come together (top graph). Each electron is attracted to the other nucleus, so Ee-n therefore decreases (middle graph). There is also a contribution to the energy from the electrons moving called the kinetic energy, Ekinetic, that is attractive at longer separations but becomes slightly positive (repulsive) near re (bottom graph). | ||
