| |||
|
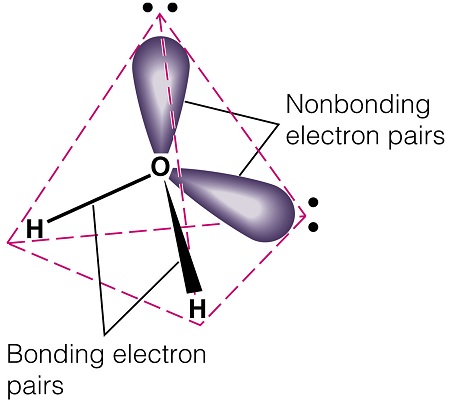
You may have selected answer (c) because you learned about the structure of water through the Valence Shell Electron Pair Repulsion (VSEPR) model. | ||
|---|---|---|
SOURCE | ||
|
VSEPR rather blithely assumes that all the electron pairs around an atom are free to orient in an optimal way to minimize pair-pair repulsion. VSEPR explicitely denies that the atom imposes any constraints on the pairs. But in fact it is undeniable that the singly occupied 2p orbitals in O are 90° apart. We think it is better to ask why the bond angle of water opens up from the nominal angle of 90° to something over 100° than to try to explain why it deviates from VSEPR's prediction of 109.5°. | ||
|
We'll follow this up later... | ||
