| |||
|
We do not cover Lewis diagrams in the module because our 2D and 3D representation for atoms include the same information but are superior: they do not omit information about the orbitals. Lewis structures discard critical information about orbital assignments and occupations. | ||
|---|---|---|
|
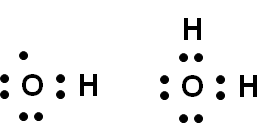
If desired, it's quite straighforward to create Lewis structures from the bonding diagrams used in the module: simply draw bond pairs and lone pairs in numbers corresponding to the quantities of each in the bonding diagram. | ||
|
Here are Lewis diagrams for OH and H2O based on the bonding diagrams in Figures 19.4 and 19.5.: | ||
| ||
|
Normally, one cannot draw a Lewis diagram for OH because the rules for Lewis diagrams rely on satisfying the octet rule, which simply doesn't apply to radicals. | ||
