|
I have been using computational techniques to study astrochemical molecules and their reactions for over a decade. In 1994 I joined a well-established computational exobiology program at Molecular Research Institute that had been started by Gilda Loew. Her efforts extended back to 1971, before the founding of MRI. My efforts followed those of Doug DeFrees and Sergio Maluendes. We collaborated with outside people including Eric Herbst, Doug MacLean, and Sheldon Green. Later, I interacted with Lou Allamandola and Max Bernstein. A bibliography (80 kb) of MRI and earlier astrochemistry papers spanning three and a half decades can be downloaded by clicking here. |

click image to enlarge |
|
Over the course of several years, I took the MRI program in a new direction: the study of organic reactions in ices. To explain why this became relevant, I will first describe why the study of interstellar molecules and their chemistry may hold great implications for the origins of life on Earth and elsewhere in the universe. | |
|
The origin of life begins with the odyssey that the elements undergo between their formation in stars by nucleosynthesis to their arrival on the surfaces of planets. In spite of the hostile conditions -- extremely cold temperatures and very low pressures -- chemical complexity can develop in cold interstellar clouds (nebulae), where over 130 molecular species have been discovered to date. These cold clouds can condense into new stellar systems in places called star-forming regions. When this happens, a protostar and its planets slowly develop. In time, most of the gas is drawn by the force of gravity into the star and its planets themselves, but some of it ends up in smaller bodies including planetoids, comets, and dust. |

click image to learn more about this star-forming region |
According to the cometary hypothesis, the raw material for prebiotic evolution on Earth may have been delivered to the surface in the form of comets, which are formed when ice-covered dust particles collect together and grow into planetoid-sized masses. Life's Far-Flung Raw Materials |
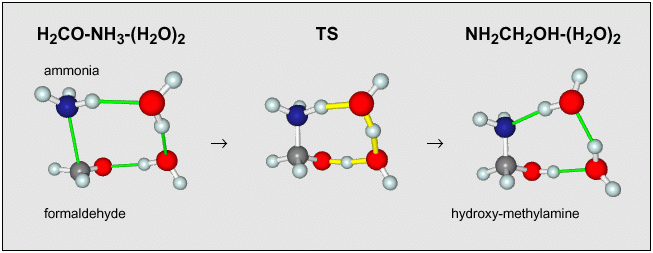
One of our ongoing projects is to characterize the manner in which ice can change the behavior of chemical reactions. As an example, the reaction between ammonia and formaldehyde has a large barrier in the gas phase, much too high to be efficient at cloud temperatures of 10-20 K. But our quantum chemical calculations found that this reaction is enhanced considerably when it occurs within an ice matrix, to the extent that there is almost no barrier remaining. The water catalyzes the reaction in two ways, by directly partipating in the reaction and by generating an electrostatic field that further reduces the barrier height. |
(2021: This mechanism was replaced by later work, when I was able to treat much larger clusters using density functional theory.) |
Now that we understand how ices can enhance some reactions, we can look for viable pathways to other organic molecules that could be precursors to prebiotic species. One of the most important classes of precursors are the amino acids, which are critical components of peptides and proteins. |

|
Visit The Astrochymist website to read more about astrochemistry. |