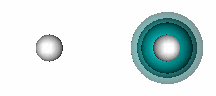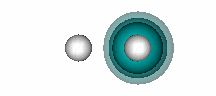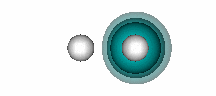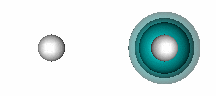| ||
|---|---|---|
| PREVIOUS SECTION | CURRENT SECTION | NEXT SECTION |
| 10. Attraction & Repulsion | 11. Combining H+/H/He | 12. Combining H with B–Ne |
| 11. Combining H+, H, and He | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
In this section we will consider H+, H, and He. We will see what we can learn by combining them two at a time. That gives us six combinations, three with the same ion or atom (H+ + H+, H + H, and He + He) and three where the ion or atom are different (H+ + H, H+ + He, and H + He). We'll work through all of these. | ||||||||||
|
Before we look at possible reactions, let's review the symbolic representations for H+, H, and He that we'll use: | ||||||||||
11.1 | 
| |||||||||
|
For H+, we will use an empty circle or a glassy sphere for its 2D and 3D representations. The H atom has a singly occupied 1s orbital, where the spin of the electron can either be up (yellow) or down (blue). The He atom has a doubly occupied 1s orbital, denoted with gray in the 3D model. | ||||||||||
|
Let's start with the H+ + H+ interaction. Here is a question for you: | ||||||||||
| ||||||||||
|
H+ + H+. The interaction of two protons involves no electrons and thus depends only on Coulomb's law. Since like charges repel, the interaction of two protons is entirely repulsive and no bond forms, answer (a) above. The figure below depicts the potential energy of two protons. The double arrow indicates that the repulsion will tend to drive the two H+ ions apart. | ||||||||||
11.2 | 
| |||||||||
|
He + He and H + He. The next two interactions we'll look at are different than the previous one, where it was straightforward to anticipate that two protons would repel each other. Now we have either two He atoms or a He and H atom approaching each other. Both H and He are neutral, so Coulomb's Law doesn't tell us what happens. The figure below shows that both of these interactions are also repulsive: | ||||||||||
11.3 | 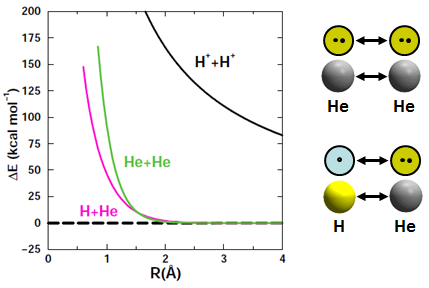
| |||||||||
|
While the He + He or H + He are both repulsive, the repulsion is much weaker and only occurs when the two particles get very close together. Since H and He are neutral, we must look elsewhere than Coulomb's Law to understand why they repel each other. | 
| |||||||||
|
The next figure is an animation that shows what happens to the orbitals for both interactions shown in Figure 11.3: | ||||||||||
11.4 |
| |||||||||
|
When the He atoms are far apart, we see their spherical 1s2 electron pairs. When the atoms get close to each other, we see that the electron pairs try to avoid each other. The 1s2 electron pairs on two He atoms repel other because of the Pauli principle. They cannot come together in the space between the two nuclei. | ||||||||||
|
The next figure shows the same orbitals as above coupled together with the potential energy curve shown in Figure 11.3 (the color change of the orbital on the left compared to the figure above doesn't mean or change anything): | ||||||||||
11.5 | 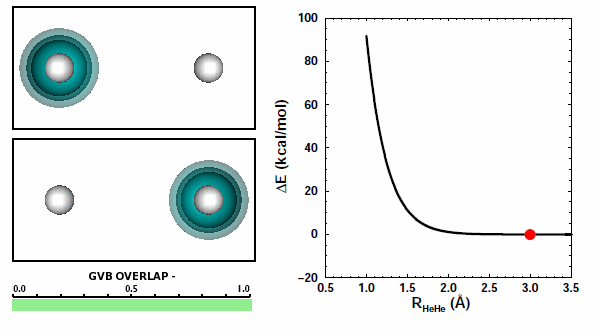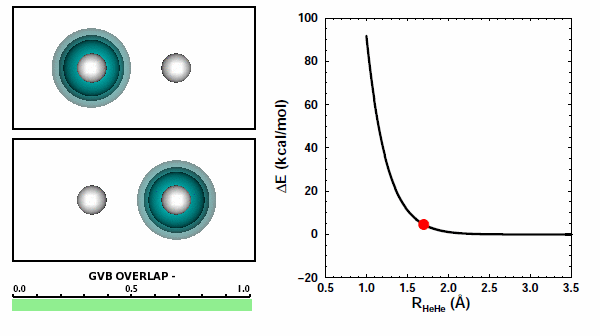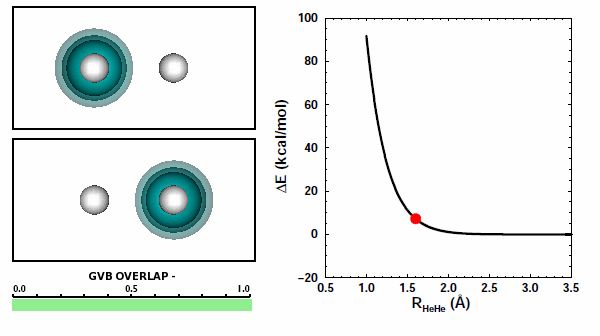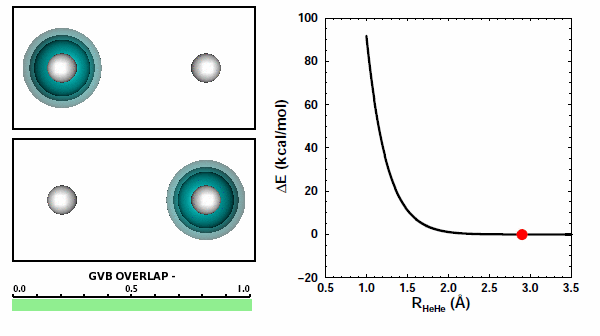
| |||||||||
|
Note that the atoms are pushing way up the steep part of the potential as they reach the nearest point they approach each other. The box is a reminder: | ||||||||||
| ||||||||||
|
H + H. The interaction between two H atoms is a bit different from the start than the three interactions we've looked at so far, which have all been repulsive. It's different because there are two unique ways that two H atoms can come together, as shown here: | ||||||||||
11.6 | 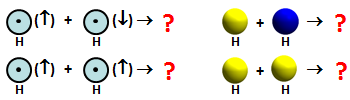
| |||||||||
|
Since the H atoms each have a singly occupied 1s orbital, they can come together either with different electron spins (top row) or with the same electron spin (bottom row). The first combination can be called antiparallel spins, and the second case is parallel spins. | ||||||||||
|
The next figure shows the two potential energy curves that arise from these combinations: | ||||||||||
11.7 | 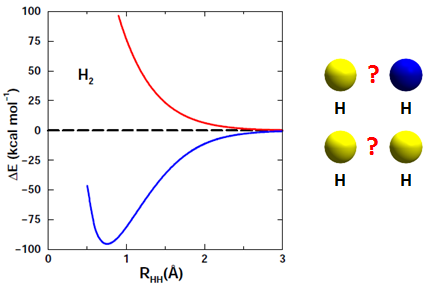
| |||||||||
|
The red curve in Figure 11.7 is another example of repulsion, but the blue curve has a minimum In Section 10 we learned that a curve like this corresponds to a bound system. We can see that the minimum falls around 0.75 Å (where 1 Å = 0.1 nm = 10-10 m) and is about -100 kcal/mol deep. The blue curve corresponds to the H2 molecule. Try to answer the following question: | ||||||||||
| ||||||||||
|
The correct answer is (b). The red repulsive curve occurs if the electrons of the two H atoms have the same spin, while the blue curve a minimum occurs if the electrons of the two H atoms have opposite spins. | ||||||||||
|
We can understand why things turn out this way if we look at what happens to the orbitals in each case. First, here are the orbitals for the repulsive case: | ||||||||||
11.8 | 
| |||||||||
|
The orbitals from the two H atoms start spherical and compress when then come together. They do not overlap at all and avoid each other. In the other case, the orbitals behave quite differently: | ||||||||||
11.9 | 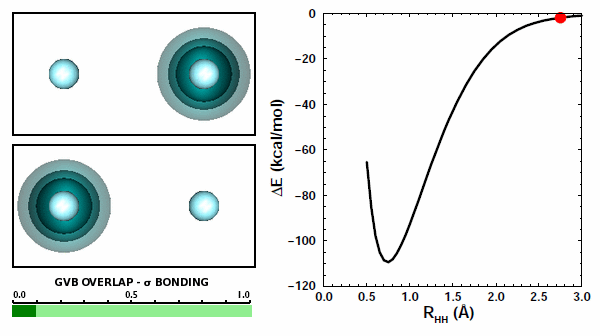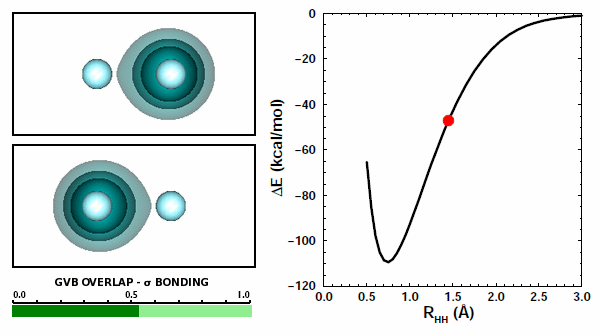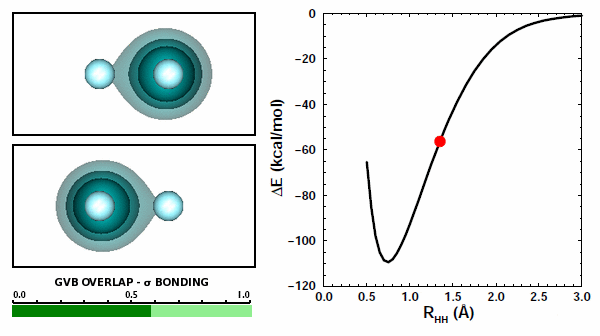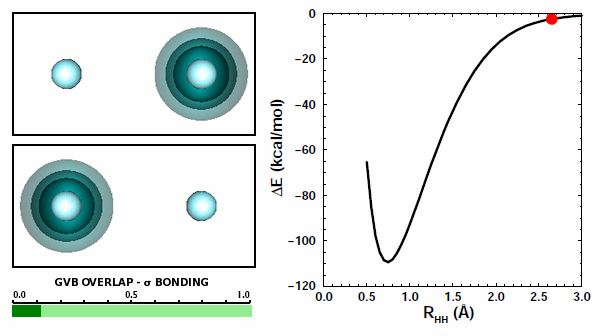
| |||||||||
|
Now the orbitals are able to overlap quite well because they are allowed to do so by the Pauli principle. The overlap increases from zero to about 0.8 at the minmimum. | ||||||||||
|
To understand why H2 is able to form, we need to go back to what we learned in the section on atoms about the way that positively and negative charged objects interact with each other. At long separations, each H atom consists of a proton and an electron that are bound together by the attractive force between the proton and electron. As the two H atoms approach each other, we have new interactions that aren't present at long separations. For example, the two protons interact with each other. The two electrons also interact with each other. You should be able to answer the following question easily: | ||||||||||
| ||||||||||
|
The correct answer is (b), of course. Both interactions are repulsive, because like charges repel each other. There is one more kind of potential interaction present in H2 that is not present in the two H atoms: each atom's electron interacts with the other atom's proton. There are two of these contributions, one from each atom. Here's something very interestin: you can see the attraction happening in the animation above. In Figure 11.9, you can see that the orbital of each hydrogen is attracted to the other nucleus and moves toward it. | ||||||||||
|
There's one more piece to the story. Let's see if you get this question right: | ||||||||||
| ||||||||||
|
Answer (c) is always wrong. Electrons never ever attract each other or bond to each other! Answer (a) is wrong for a similar reason: an electron and a proton always attract each other, but that's not enough to let H2 form when the electron spins are the same. The correct answer is (b): H2 can form in the case when the electrons have opposite spins because the Pauli principle lets them overlap. | 
| |||||||||
|
We call the type of bond that forms in H2 a covalent bond because each atom contributes one electron to the bond, and they are shared equally by the two nuclei. Here is H2 formation in symbolic form: | ||||||||||
11.10 | 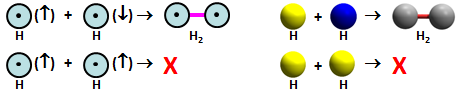
| |||||||||
|
The pink bar between the orbitals in the 2D model and red rod between the spheres in the 3D model indicates the bond. In the 3D model, we also changed the color from yellow (up) and blue (down) to gray (antiparallel pair). We call this pairing of electrons coupling the electrons, as in, "a covalent bond is formed in H2 when the electrons in two H 1s orbitals are coupled together spin up and spin down." Again, this does not mean there is any attraction between the electrons that are coupled together! | ||||||||||
|
Let's summarize what we've learned about chemical bonding with a box: | ||||||||||
| ||||||||||
|
Although many chemical bonds occur because atoms or molecular fragments each contribute one electron, it turns out that's not the only way that a bond can form. We will see two exceptions when we explore the interactions between H+ and either H or He. | ||||||||||
|
H+ + H. The figure below shows that a bond forms when H approaches H+. The bond is longer than the one in H2 by about 0.25 Å, and it is somewhat weaker (about two-thirds as strong). | ||||||||||
11.11 | 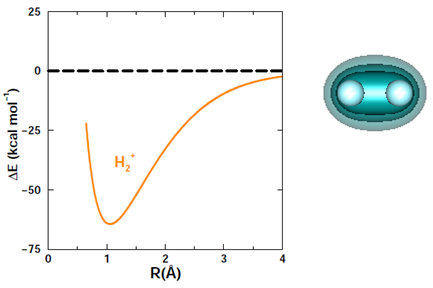
| |||||||||
|
In spite of the differences, H2+ is clearly a bound molecule. There is only one electron in H2+, so it is clearly not essential to have a pair of electrons to form a bond (although one-electron bonds are quite rare). The orbital for the single electron is shown above. It is shared equally between the two nuclei. The two protons repel each other, but their mutual attraction to the lone electron is sufficient to hold the three particles together. | ||||||||||
|
H+ + He. The figure below shows that a bond also forms when He and H+ interact. | ||||||||||
11.12 | 
| |||||||||
|
In HeH+, there are two electrons involved in the bond, but they both originate bound to the He atom. H+ has an empty 1s orbital. It attracts the 1s2 pair of electrons of He toward itself as it approaches He, as shown in the animation below (the He nucleus is on the left, the H nucleus on the right): | ||||||||||
11.13 |
| |||||||||
|
The bonding in HeH+ is different from the covalent bonding in compounds like H2 because both electrons in the shared pair originate on one of the atoms (He, in this case). This type of bond is known as a dative bond. The other atom (or ion, in this case) can participate in the bond because it has an unoccupied orbital that allows its nucleus to attract the pair of electrons on He without any interference from a singly or doubly occupied orbital. Remember that we saw above that when He interacts with H, the potential energy curve was repulsive (Figure 11.3). | ||||||||||
|
We will see other cases where H+ can interact with an electron pair to form a dative bond in molecules like H3O+ (hydronium ion) and NH4+ (ammonium ion). There are also other atoms than can have an unoccupied orbital that lets them form dative bonds. | ||||||||||
|
Bond polarization. In H2 and H2+ the electron(s) are shared equally between the two H nuclei. We will see other cases where the sharing is equal, but there are also a lot of cases where the sharing is not equal. The bond in HeH+ is an example of this. While the H+ attacts the He 1s2 orbital toward itself, much of the orbital remains on He. We call this unequal sharing a polarized bond. When a bond is polarized, there are different charges on the two fragments, and those unlike charges result in a property known as the dipole moment. | ||||||||||
|
To summarize, in this section we explored the six combinations of pair interactions drawn from the very small set of H+, H, and He and found three different types of bonding. Here is a list of bond types identified so far: | ||||||||||
| ||||||||||
|
Just as important, we also observed a number of cases where interactions were repulsive, including H+ + H+, H + He, He + He, and H + H when the spins of the electrons are the same. | ||||||||||
|
In the next section, we will explore what happens when H interacts with atoms that have 2p electrons, include boron through neon. | ||||||||||
| Click on the link to proceed to the next section: | 12. Combining H with B–Ne |
|---|---|
| MODULE TABLE OF CONTENTS | |


|
| Copyright 2011-2017 University of Illinois. All rights reserved. |
|---|